Super-Resolution microscopy allows insights into intracellular processes and interactions at a molecular level. During the past two decades, advances in the field have led to significantly higher resolved images, faster processing times, and further insight into the functionality of molecular machines within cells. We discuss how nanometer precision measuring and positioning technology has been a major enabler for rapid advances in this field, and the importance of nanopositioning for Super-Resolution microscopy applications.
Background
The development of super-resolution microscopy (SRM) has led to renewed interest in the use of optical microscopy in a number of areas of life sciences research, especially neuroscience and molecular cell biology. First proposed in 1978, this technique overcomes the so-called Abbe diffraction limit – described by German physicist Ernst Abbe over 100 years earlier in 1873 – allowing structures of less than 200 nanometers to be distinguished with fluorescence-based light microscopy.
Super-Resolution microscopy remained a niche technique until around 15 to 20 years ago, when researchers began combining a number of separate super-resolution technologies – including light sheet, 4Pi, STED (stimulated emission depletion), PALM (photoactivation localization microscopy), and STORM (stochastic optical reconstruction microscopy) – to create systems capable of achieving spatial resolutions down to around 20 nanometers. This development led to Eric Betzig, Stefan W. Hell, and William E. Moerner being awarded the 2014 Nobel Prize in Chemistry, and has greatly enhanced our understanding of many intracellular processes and molecular interactions.
SRM allows cell biologists to non-destructively investigate cellular structures in previously unattainable detail, and its use has grown rapidly, particularly since the arrival of commercially-available super-resolution systems.
One of the main benefits of using these optical microscopy systems is that, unlike other methods offering comparable resolution – such as scanning electron microscopy (SEM) – they can be used to investigate metabolic interactions in living cells. The rise of SRM has therefore prompted many laboratories to revisit imaging-based research in areas such as cell division and intracellular transport – which had previously been hindered by diffraction-limited image resolution – by combining selective labelling with the ability to distinguish co-localised molecules of interest using spectrally-separated fluorescent dyes.
As with many new research tools, the development of SRM technologies has been primarily driven by academia with a majority of the systems in use today originating either directly from research labs or via university spin-off companies. The first generation of these instruments was, by necessity, built in-house by physicists using standard parts from other microscope platforms.
Given the 10-fold improvement in resolution achieved by SRM techniques, the ability to position samples and objectives accurately was obviously a key requirement to be able to create such systems. Fortunately, piezo-based positioning systems – with nanometer resolution – were already commercially available when SRM development began, and were widely used by other high resolution imaging techniques, such as atomic force microscopy (AFM) and SEM. As a result, piezo-based nanopositioners became a key enabler for the development of SRM techniques.
The Piezo Effect
Piezo – derived from the Greek word for pressure – technology was discovered in 1880 by brothers Jacques and Pierre Curie, after they identified that applying pressure to crystals, such as quartz and tourmaline, generated an electrical charge.
This effect constitutes conversion of mechanical energy into electrical energy – coined the ‘direct piezoelectric effect’ – in crystals and ceramics with non-symmetric elementary cells below the Curie temperature. The pair went on to discover that conversely, applying an electric field to certain crystals can deform the material, resulting in a change of its length – the inverse piezoelectric effect.
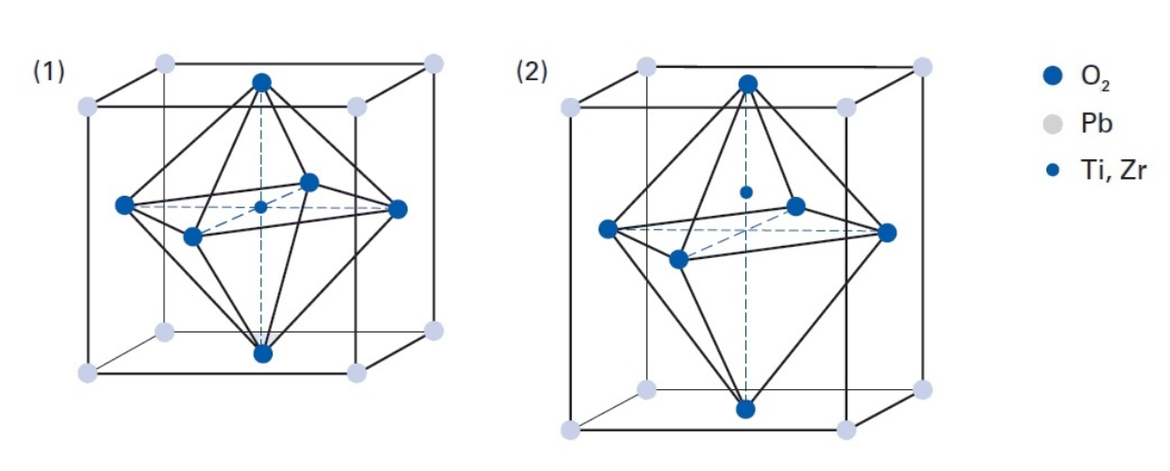
The piezoelectric effects in natural monocrystalline materials, such as the afore-mentioned quartz and tourmaline, are relatively small. However, engineered polycrystalline ferroelectric ceramics – including barium titanate (BaTiO3) and lead zirconate titanate (PZT) – display much larger displacements or induce larger electric voltages. These piezoceramic materials can also be doped with ions – for example Ni, Bi, La, Nd or Nb – to optimize the piezoelectric effects and parameters, allowing each piezo component to be individually adapted according to the intended application.
Practical Applications
Piezoelectric actuators have long been considered the gold standard for high precision positioning of both sample stages and objectives in a broad range of microscopy techniques. The main advantage of this technology is that movement is controlled by directly converting electrical energy into mechanical motion through solid state effects. In theory, the piezoceramic material itself has unlimited resolution, allowing precise, stable, and reproducible fine positioning down to the sub-nanometer range, and typical travel ranges between ten and a few hundred micrometers for microscopy-related applications such as sample stages or Z-focusing drives.
Despite traditional light microscopes not requiring this level of precision, many of these products have been in widespread use for over 20 years. This is thanks to their combination of positional accuracy, high speed movement, and improved stability compared to motorized drives.


As piezo materials have no traditional moving mechanical parts, the resulting motion is completely friction free and not subject to wear, making them virtually maintenance free and capable of several billion cycles without measurable changes in their behavior. They are also both vacuum and clean room compatible.
Another positive characteristic of piezo ceramic material is its extremely rapid response to a drive voltage change along with acceleration rates of thousands of g’s. In real world applications, piezo-flexure-driven nanopositioning stages with travel ranges in the 100µm to 500µm range achieve step and settle times from 5 to 20 milliseconds, while long travel stages with ultrasonic piezo motors can reach velocities up to 100’s of millimeters per second, making them ideal for fast scanning and high throughput microscopy applications.
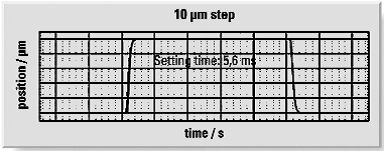
Conversely, piezoceramics can be implemented in standing and travelling wave ultrasonic motors for microscopy stages with travel ranges of 100mm and above or rotation stages with unlimited rotation. These motors provide a very wide dynamic velocity range from less than one micron per second to 100’s of mm per second. This proven track record across multiple microscopy disciplines has made piezo actuators and motors the natural choice for laboratories developing the first generation of SRM platforms; they were both familiar to researchers and already offered the performance required.

Nanometers Ahead
It may seem serendipity that these ceramic actuators already in widespread use offered resolution almost two orders of magnitude higher than was required for existing light microscopy applications, but the other characteristics of this technology – exceptional stiffness, excellent reproducibility, and extremely rapid movement – made it the logical choice. The advent of SRM has now allowed optical microscopy to fully exploit the capabilities of these existing electro-ceramic elements.
High-resolution microscopy techniques must be capable of accurate and reproducible positioning of imaging elements or samples. Applications range from precise Z-focus positioning of light microscope objectives in the depths of field to exact positioning of tiny biological samples for 4Pi Resolft 2-Photon super-resolution fluorescence microscopy. Also in AFM, SEM, and TEM, as well as correlative microscopy, versatile nanopositioning solutions greatly support even the most demanding positioning tasks.
Piezo mechanisms are currently used for three main applications in SRM: Z-positioning of objectives for autofocusing; manipulation of illumination systems (such as in light sheet and 4Pi techniques); and for sample positioning. Each of these applications obviously has unique requirements in terms of range of travel, reproducibility, and settling times but, in general terms, system integrators want piezo-controlled movements to be as rapid, precise, and stable as possible.
Why Speed Matters
The exceptional resolutions which can be achieved using SRM – and resulting decrease in the field of view – has naturally led to a significant increase in the number of images required to complete a full scan of even a single plane of an individual cell. For other techniques offering similar image resolution, for example SEM, this increase in the number of images required, and subsequently the time taken per scan, is not a significant issue, as they can only be used with dead cells or other inanimate materials, which will not change over the course of the scan.
In contrast, SRM allows metabolic processes to continue throughout image acquisition. The attributes, and resulting speed, of the piezo actuators therefore directly influence the quality of experimental results as the faster a scan can be completed, the better it represents a ‘snapshot’ of a single time point. Add to this the ability to scan through the entire Z stack to create a 3D render of a cell or tissue and any improvements that can speed up imaging – longer travel for sample stage movement, reduced drift, shorter settling times, etc. – become increasingly important.
Finding Your Molecule Again
When performing quantitative image analysis, it is essential that users can bookmark the position of cells or structures of interest then quickly and consistently relocate those same structures at a later time or on a different instrument. This is particularly important for longitudinal (time-lapse) studies to understand metabolic, molecular, and information transport on a cellular level, as these processes often have very short timescales.
Bidirectional positioning reproducibility – the precision with which the system can return to a specific X and Y position – is therefore becoming increasingly important, and piezo-nanopositioners can reliably move the sample or objective to the correct place consistently and precisely. By allowing the motion of the stage to be measured directly with a capacitive sensor – without any influence from the drive or guide elements – this set-up ensures optimum repeatability, outstanding stability and stiffness, and fast-responding control.
Total internal reflection fluorescence microscopy (TIRFM) is a comparatively simple and inexpensive super-resolution fluorescence microscopy method that has been used for many years, and TIRFM systems – such as the GATTAscope – benefit greatly from piezo-based solutions. For this technique, a laser beam hits the glass surface of a microscope slide, microplate or Petri dish at a total internal reflection angle to stimulate fluorescence.
Since the electromagnetic field of this beam only penetrates the sample on the other side of the glass plate to a depth of 100 to 200 nm, the stimulated area in the Z plane is reduced, offering better Z resolution than standard wide field or confocal fluorescence microscopy. Piezo solutions enable the precise positioning of lasers and Z-drive that are key to successful super-resolution imaging in the Z-direction.
Similarly, light sheet fluorescence microscopy – as used by the Janelia research group2 – uses a unique planar illumination approach, orthogonal to the observation direction. This minimizes photobleaching and phototoxicity for living specimens, allowing 3D and live cell imaging of large specimens over an extended period of time.
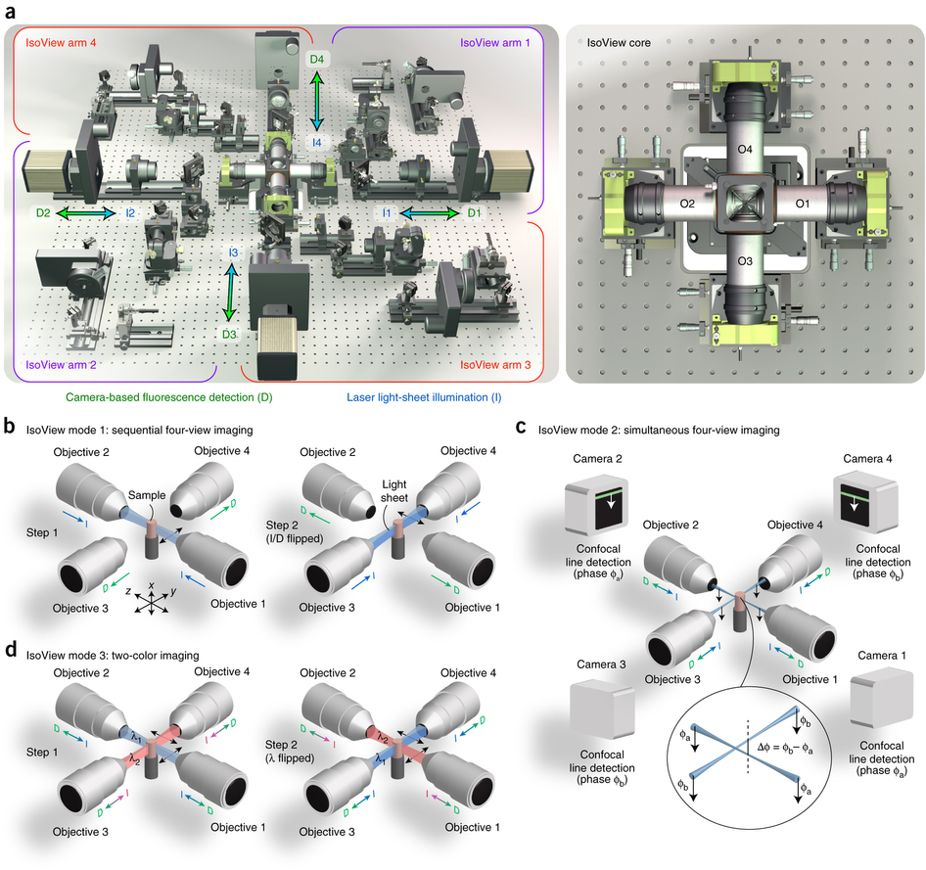
Schematic representation of the IsoView light sheet fluorescence microscope.
Left: (A) four orthogonal arms provide simultaneous light sheet illumination and fluorescence detection. The specimen is located at the center of this arrangement, and volumetric imaging is performed by sweeping light sheets across the sample with objective piezo positioners.
Right: close-up view of sample chamber showing objectives (O1-O4) and piezo positioners.
In mode 1 (B), four views are acquired by alternating light sheet illumination (l) and fluorescence detection (d) along orthogonal axes, with temporal separation used to avoid cross-talk between views.
In mode 2 (C), illumination and detection are performed at the same time in all arms, and cross-talk is avoided via spatially matched beam scanning and confocal detection using a phase offset in orthogonal arms (spatial separation).
In mode 3 (D), different illumination wavelengths are used in orthogonal arms simultaneously, and four views are acquired for both channels by alternating wavelengths (spectral separation).
Examining small organisms in this way requires samples to be positioned to within a range of a few millimeters in XY with a bidirectional repeatability of 100 nm, which can be achieved efficiently with piezo-driven stages and objectives.
The high throughput nature of piezo-based stages allows imaging of large-surface objects – for example high magnification image recording of objective slides and micro well plates– within seconds. During image recording, piezo-controlled stages precisely and continuously move the object, either in ‘stop-and-go’ mode, or at a constant velocity in ‘fly-over’ mode. It is also possible to combine this scanning process with real-time data handling and image-processing steps to ensure that even CPU-intensive tasks, such as the superimposed tiling processes, run with limited delay. This allows individual images to be merged seamlessly into the overall image even while acquisition is in progress, while continuously adjusting the focus during scanning.
Summary
Super-resolution microscopy is driving rapid advances in our understanding of many cellular processes and molecular interactions. The latest generation SRM systems are achieving spatial resolutions of around 20 nanometers, and the sub-nanometer positioning precision offered by current piezo-based drives will help to increase this resolution even further.
Despite this, microscope integrators have been demanding longer travel ranges, greater stiffness, and higher speeds since the construction of the very first SRM systems, and the development of customized solutions to meet this growing demand will be vital as optical microscopy pushes further into the nano-world.
Blog Categories
- Aero-Space
- Air Bearing Stages, Components, Systems
- Astronomy
- Automation, Nano-Automation
- Beamline Instrumentation
- Bio-Medical
- Hexapods
- Imaging & Microscopy
- Laser Machining, Processing
- Linear Actuators
- Linear Motor, Positioning System
- Metrology
- Microscopy
- Motorized Precision Positioners
- Multi-Axis Motion
- Nanopositioning
- Photonics
- Piezo Actuators, Motors
- Piezo Mechanics
- Piezo Transducers / Sensors
- Precision Machining
- Semicon
- Software Tools
- UHV Positioning Stage
- Voice Coil Linear Actuator
- X-Ray Spectroscopy